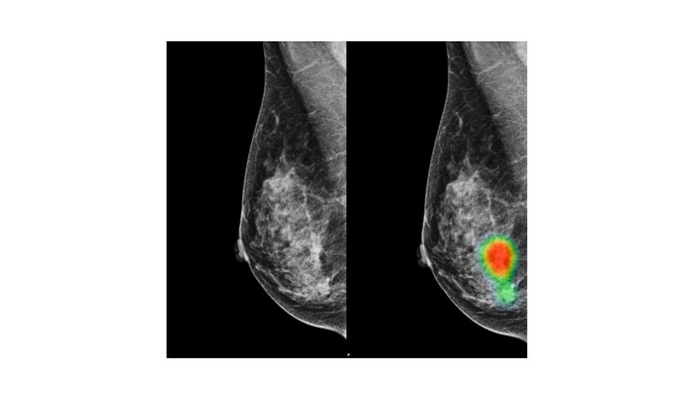
Lunit, a medical AI company specializing in developing AI solutions for radiology and oncology, announced that its AI solution for breast cancer, Lunit INSIGHT MMG, has been CE certified. The medical software, which was certified on 26 May, is now commercially available across the European Economic Area.
Lunit INSIGHT MMG was first introduced at the 2018 Radiological Society of North America (RSNA) annual meeting and analyzes mammography images with 97% accuracy, providing the location of lesions suspicious for breast cancer. It also provides an abnormality score that reflects the probability of the existence of detected lesions.
The company was founded in 2013 and Lunit INSIGHT MMG is its second product after its INSIGHT CXR product was launched in 2017.
HIMSS20 Digital
THE LARGER TREND
In February this year, a study by Korean hospitals and Lunit which was published in Lancet Digital Health demonstrated the benefits of AI-aided breast cancer detection from mammography images. One of the major findings showed that AI, in comparison to the radiologists, displayed better sensitivity in detecting cancer with mass (90% vs 78%) and distortion or asymmetry (90% vs 50%), Healthcare IT News reported.
In late 2019, received a CE mark for its AI solution for chest x-ray, Lunit INSIGHT CXR. According to the company, Lunit INSIGHT CXR has been deployed to assist the diagnosis of COVID-19 patients from Daegu in South Korea and nearby regions, the hardest-hit area where approximately 8,000 patients or about 85% of the entire domestic cases (around the period of mid-March 2020) — has been diagnosed.
ON THE RECORD
“Among the patients suspicious of breast cancer upon screening mammography, only 29% is actually diagnosed with cancer after a biopsy. I am delighted to introduce Lunit INSIGHT MMG, now CE certified, to healthcare professionals and institutions across the continent of Europe. With our AI software, we would like to increase the effectiveness of mammography screening and reduce unnecessary biopsies,” said Brandon Suh, CEO of Lunit.
